Research Interests
Nucleophilic Organocatalysis
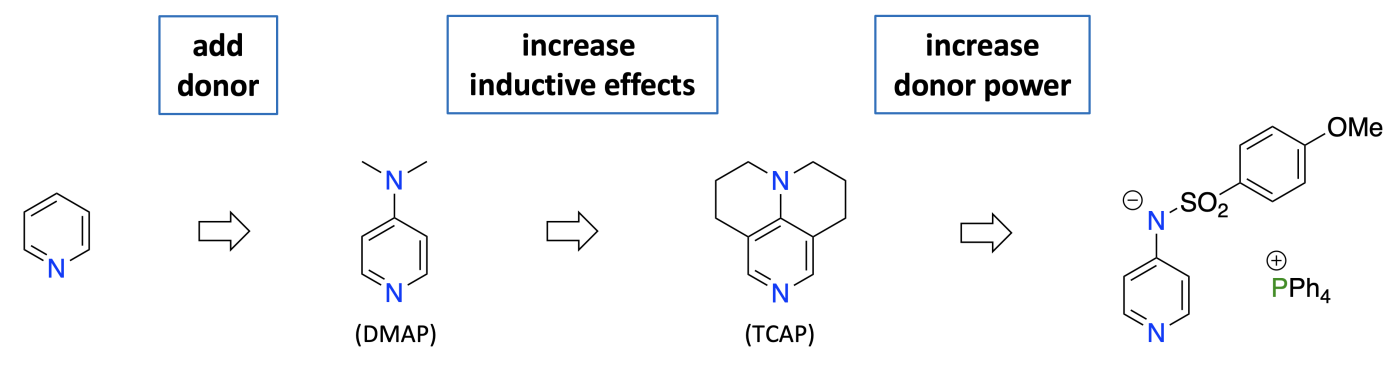
Nucleophilic catalysts are of outstanding importance in synthetic protocols for the synthesis of ethers, esters and amides. Using a combination of experimental and theoretical techniques, we develop new catalysts of enhanced Lewis basicity and nucleophilicity. The latest iteration of these efforts centers around ion pair catalysts, whose modular structure has opened new avenues into catalyst design.
selected publications
"Pyridinamide Ion Pairs: Design Principles for Super-Nucleophiles in Apolar Organic Solvents",
V. Burger, M. Franta, A. C. O‘Donoghue, A. R. Ofial, R. M. Gschwind, H. Zipse, J. Org. Chem. 2025, 90, 2298 - 2306. [DOI: 10.1021/acs.joc.4c02668]
"Highly Nucleophilic Pyridinamide Anions in Apolar Organic Solvents due to Asymmetric Ion Pair Association",
V. Burger, M. Franta, A. R. Ofial, R. M. Gschwind, H. Zipse, J. Am. Chem. Soc. 2025, 147, 5043 - 5050. [DOI: 10.1021/jacs.4c14825]
"Pyridinyl Amide Ion Pairs as Lewis Base Organocatalysts",
J. Helberg, T. Ampßler, H. Zipse, J. Org. Chem. 2020, 85, 5390 - 5402. [DOI: doi/10.1021/acs.joc.0c00114]
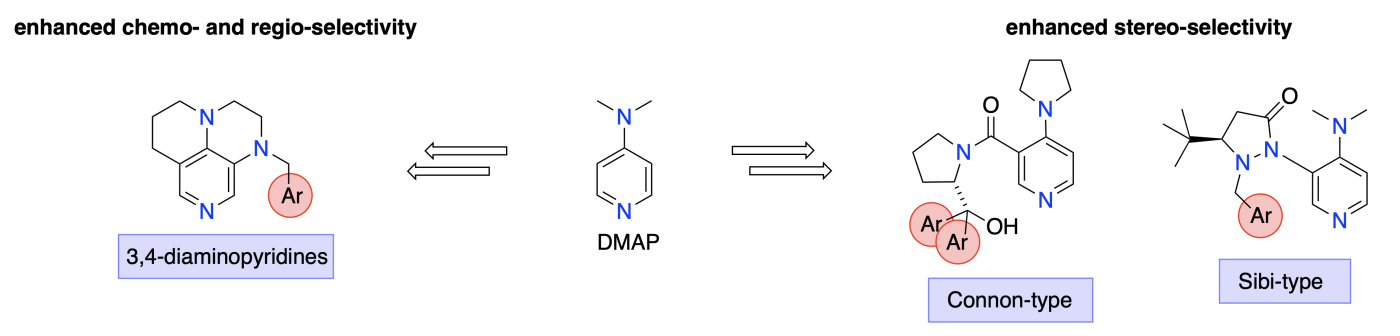
Optimization of the substitution pattern of donor-substituted pyridines provides a systematic strategy for the development of chemo-, regio- and stereoselective Lewis base catalysts. This is of particular relevance for selective transformations of polyol substrates, where combined catalyst- and reagent-variations lead to best results.
selected publications
"Size and Polarizability as Design Principles for Stereoselective Catalysis",
C. M. Carter, J. Brossette, H. Zipse, Chem. Eur. J. 2025, 31, e02741. [DOI: 10.1002/chem.202502741]
"Kinetic Resolution of Secondary Alcohols Using Connon Type Catalysts",
C. M. Carter, H. Zipse, Eur. J. Org. Chem. 2025, 28, e202500733. [DOI: 10.1002/ejoc.202500733]
"Annelated Pyridine Bases for the Selective Acylation of 1,2-Diols",
S. Mayr, H. Zipse, Eur. J. Org. Chem. 2022, e202101521. [DOI: 10.1002/ejoc.202101521]
Radical Chemistry in the Biological Context
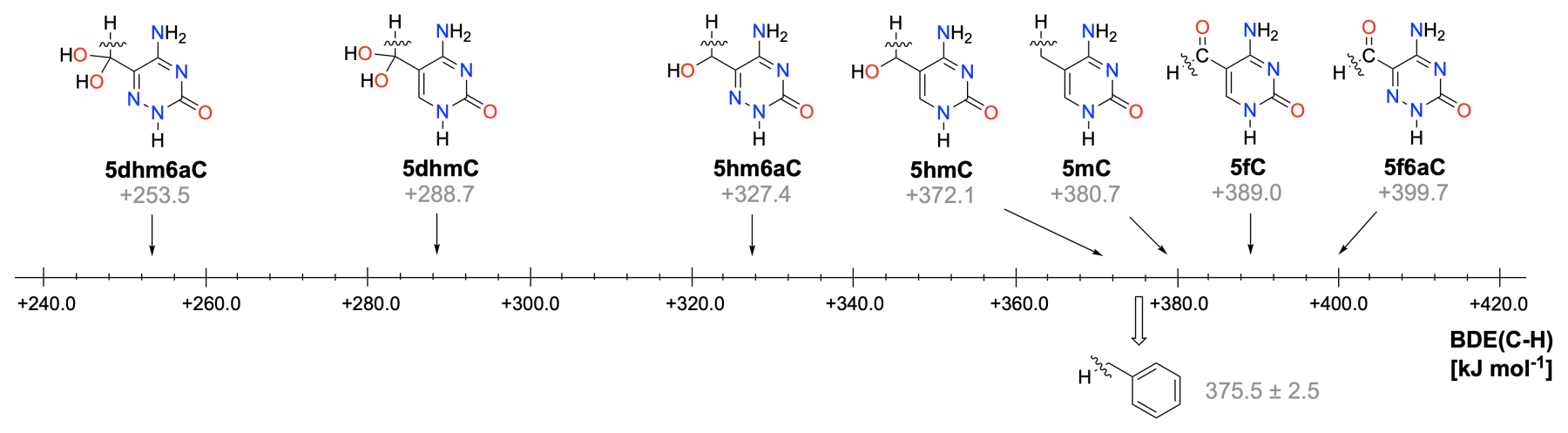
Radical chemistry in the biological context differs from that used in synthetic organic chemistry in the choice of reaction medium (water vs. low-polarity organic solvents) as well as the underlying reaction mechanisms (non-chain- vs. chain-reactions). Despite these fundamental differences, radical reactivity is in both cases guided by general concepts such as radical stability and radical polarity. The quantitative assessment of these properties with appropriately chosen theoretical methods allows not only the rationalization of experimental observables, but also predictions for new systems. This becomes especially powerful when combined with microkinetics simulations of reaction outcomes.
selected publications
"50 Years of Giese Reaction – a Personal View",
M. Spichty, H. Zipse, S. Majouri, K. M. Fromm, B. Giese, Angew. Chem. Int. Ed. 2026, 65, e24825. [DOI: 10.1002/anie.202524825]
"Aldehyde or Hydrate? Investigation into the Oxidation of 5-Formylcytosine Derivatives Using a Computational and Experimental Approach",
K. Liu, A. Menke, F. L. Zott, D. Mayer, L. J. Daumann, H. Zipse, ChemBioChem 2025, 26, e202500480. [DOI: 10.1002/cbic.202500480]
"Redox-Mediated Amination of Pyrogallol-Based Polyphenols",
S. S. Ashirbaev, N. F. Bras, P. Frei, K. Liu, S. Moser, H. Zipse, Chem. Eur. J. 2024, 30, e202303783. [DOI: 10.1002/chem.202303783]